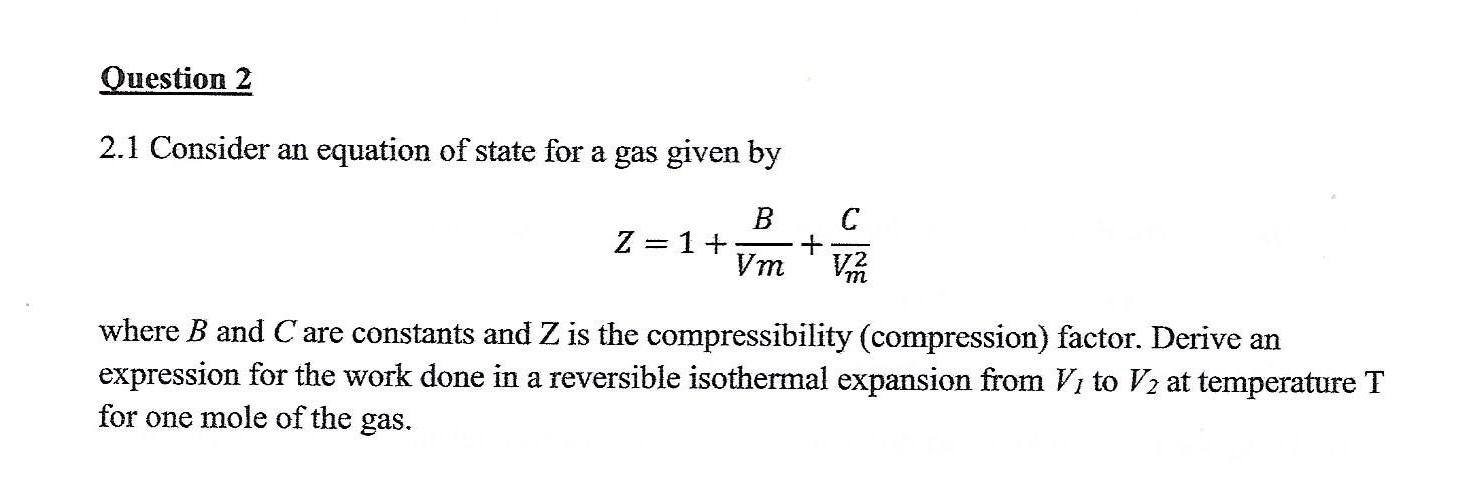SOLVED: Derive an expression for the compression factor of a gas
SOLVED: Derive an expression for the compression factor of a gas- Solved Define the compression factor, Z and explain its
 the compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0.5
the compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0.5 If `Z` is a compressibility factor, van der Waals' equation at low
If `Z` is a compressibility factor, van der Waals' equation at low Telugu] What is compressiblity factor?
Telugu] What is compressiblity factor? Show that the van der Waals equation leads to values of Z <
Show that the van der Waals equation leads to values of Z < Compressibility Factor
Compressibility Factor- At Critical Temperature,pressure and volume . The compressibility Factor (Z) Is
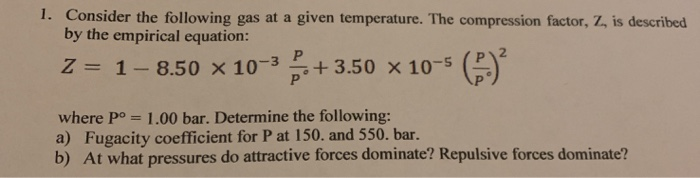 Solved 1. Consider the following gas at a given temperature.
Solved 1. Consider the following gas at a given temperature.- What is the compression ratio, and how is it calculated? - Quora
 53 pts!! The function f(x)= 7^x+1 is transformed to function g through a horizontal compression by a factor
53 pts!! The function f(x)= 7^x+1 is transformed to function g through a horizontal compression by a factor UNUB At Boyle temperature, the value of compressi factor Z has a
UNUB At Boyle temperature, the value of compressi factor Z has a SOLVED: Derive an expression for the compression factor of a gas that obeys the equation of state p(V - nb) = nRT, where b and R are constants. If the pressure and
SOLVED: Derive an expression for the compression factor of a gas that obeys the equation of state p(V - nb) = nRT, where b and R are constants. If the pressure and Compression Factor Exam Problem using Molar Volumes - Fully Explained!
Compression Factor Exam Problem using Molar Volumes - Fully Explained! a) Suppose that $10.0\ \mathrm{mol}\ \mathrm{C}_{2} \mathrm
a) Suppose that $10.0\ \mathrm{mol}\ \mathrm{C}_{2} \mathrm Solved) - For values of z near 1, it is a good approximation to
Solved) - For values of z near 1, it is a good approximation to Solved The virial expansion of the compression factor (Z)
Solved The virial expansion of the compression factor (Z) Compared with the graph of the parent function, which equation shows only a vertical compression by a
Compared with the graph of the parent function, which equation shows only a vertical compression by a Write an equation for the transformation of y=x vertical compression by a factor of 1/11
Write an equation for the transformation of y=x vertical compression by a factor of 1/11 Pick only the incorrect statement.for gas A, a=0,the compressibility factor is linearly dependent on pressure.for gas C,aneq 0,bneq 0,it can be used to calculate a and b by giving lowest P value.for
Pick only the incorrect statement.for gas A, a=0,the compressibility factor is linearly dependent on pressure.for gas C,aneq 0,bneq 0,it can be used to calculate a and b by giving lowest P value.for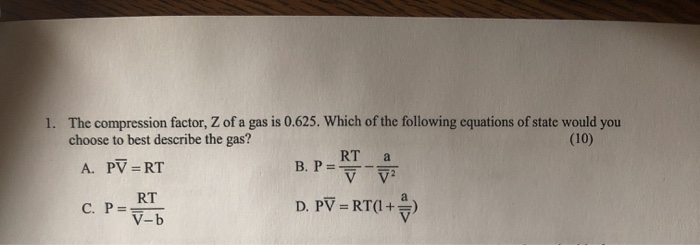 Solved 1. The compression factor, Z of a gas is 0.625. Which
Solved 1. The compression factor, Z of a gas is 0.625. Which The compression factor (compressibility factor) for 1 mol of a van der
The compression factor (compressibility factor) for 1 mol of a van der- Solved Question 2 2.1 Consider an equation of state for a
 Find the isothermal compressibility `x` of a Van der Walls gas as a function of volume
Find the isothermal compressibility `x` of a Van der Walls gas as a function of volume